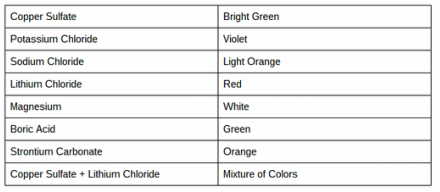
My friends and I did a lab where we burn certain salts or metals or a combonation of both and see what colors would appear. Heres a chart that we made when we recorded our findings . Something that I hypothosised about is that the colors depended on how much oxygen and electrons were in the salt/metal.
Captions for the Pictures:
Green Flame: Copper Sulfate
Red Flame: Lithium Chloride
Mix of Colors: Both
Bright light: Magnisium
Captions for the Pictures:
Green Flame: Copper Sulfate
Red Flame: Lithium Chloride
Mix of Colors: Both
Bright light: Magnisium
I saw that each element reacted to the heat from the bunsin burner, heat speeds up electrons and give energy to them and they do not know what to do with the extra energy. The color was the elements coming into view. This lab can lead into the creation and manafacturing of color changing candles or bonfire color changing packets. This lab was really mesmerizing becasuse the constant color changes and personally I am easily amused, the constant visual stimulation was really engaging.
 RSS Feed
RSS Feed
